
Tim Collett of the United States Geological Survey (USGS) proposed pumping carbon dioxide into subsurface methane clathrates, thereby releasing the methane and storing the carbon dioxide. 
On Earth, CO 2 hydrate is mostly of academic interest. A river-bed-like outflow channel can be seen, originating from Iani Chaos and extending towards the top of the image. In this mosaic taken by the Mars Global Surveyor: Aram Chaos - top left and Iani Chaos - bottom right. The CO 2 hydrate was classified as a Type I clathrate for the first time by von Stackelberg & Muller (1954). Takenouchi & Kennedy (1965) measured the decomposition curve from 45 bars up to 2 kbar (4.5 to 200 MPa). Tamman & Krige measured the hydrate decomposition curve from 253 K down to 230 K in 1925 and Frost & Deaton (1946) determined the dissociation pressure between 273 and 283 K (0 and 10☌). Three years later, he published the hydrate dissociation curve in the range 267 K to 283 K (-6 to 10☌). Villard deduced the hydrate composition as CO 2 (from French)" This already indicates the importance of the surface available for reaction (i.e. He also mentions that ".the hydrate is only formed either on the walls of the tube, where the water layer is extremely thin or on the free water surface. He was the first to estimate the CO 2 hydrate composition, finding it to be approximately CO 2 He noted that gas hydrate was a white material resembling snow and could be formed by raising the pressure above a certain limit in his H 2O - CO 2 system. The first evidence for the existence of CO 2 hydrates dates back to the year 1882, when Zygmunt Florenty Wróblewski reported clathrate formation while studying carbonic acid. It is also quite likely to be important on Mars due to the presence of carbon dioxide and ice at low temperatures. CO 2 hydrates are widely studied around the world due to their promising prospects of carbon dioxide capture from flue gas and fuel gas streams relevant to post-combustion and pre-combustion capture. The clathrate can exist below 283K (10 ☌) at a range of pressures of carbon dioxide. There has also been some experimental evidence for the development of a metastable Type II phase at a temperature near the ice melting point. ( June 2012) ( Learn how and when to remove this template message)Ĭarbon dioxide hydrate or carbon dioxide clathrate is a snow-like crystalline substance composed of water ice and carbon dioxide. Please help improve it to make it understandable to non-experts, without removing the technical details. It is flammable.This article may be too technical for most readers to understand. 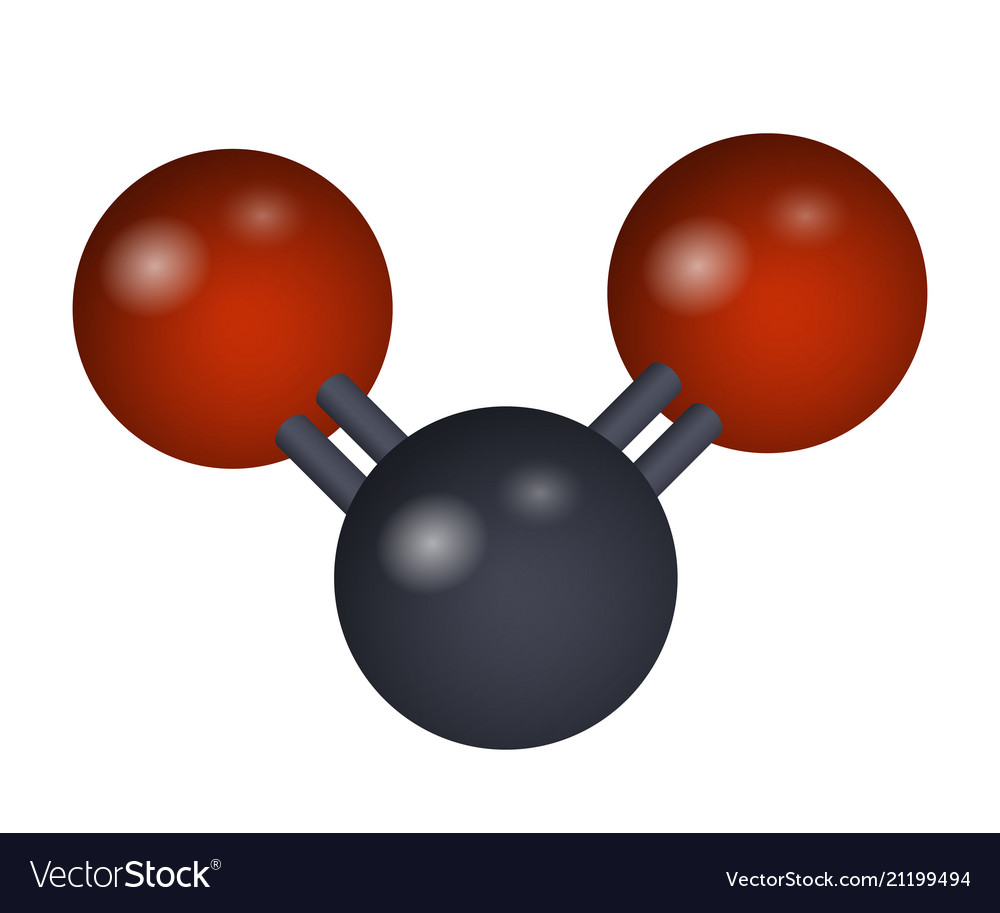
Carbon monoxide also ignites easily, causing flame. It replaces the position of the oxygen in the haemoglobin, producing carboxyhemoglobin causing asphyxia. This gas is extremely toxic and causes dozens of fatal air poisoning. Health effects / safety hazards: Carbon monoxide is is fatal by inhalation. Carbon monoxide is also present in some mixtures of fuel. Other uses include the formation of chemical intermediaries through reactions that use carbon monoxide or end products as methanol, ethylene and acrylates. It is also a reagent in some organic synthesis reactions as the Frischer-Tropsh process. Uses: Carbon monoxide can be used in chemical industry as a reducing agent. This fact along with the similar size between both atoms also contributes with the stability and low reactivity of the molecule. The strong dipole moment due to the difference of electronegativity between the oxygen and carbon create a partial charge of -1 over the oxygen and +1 over the carbon atom. It is largely soluble in water and other solvents as chloroform, acetic acid, ethanol, methanol, toluene and ammonium hydroxide.Ĭhemical properties: Carbon monoxide is a stable molecule due to the strength exhibits by the triple bond. Its melting point is -205 ☌ and the boiling point is -191.5 ☌. The density of this gas is 0.79 g/mL (liquid) and 1.145 g/mL (gas). Physical properties: Carbon monoxide is a colourless, odourless gas. Carbon monoxide can also be produced by the oxidation of hydrocarbon gases that are present in the natural gas. Through these processes is produced all the carbon monoxide found in atmosphere. Preparation: Carbon monoxide is produced from the incomplete combustion of carbon, the decomposition of organic compounds or though the reduction of carbon dioxide. Carbon monoxide is also present in volcanoes. Occurrence: Carbon monoxide is found in nature, as produced in low concentrations by some archaea and bacteria. 
Its chemical structure can be written as below, in the common representations used for organic molecules. As in all triple bonds, it is formed by one sigma bond and two pi bonds and the molecule acquire a linear geometry. The molecule is formed one carbon atom and one oxygen atom joined by one triple bond. Formula and structure: The carbon monoxide chemical formula is CO.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
